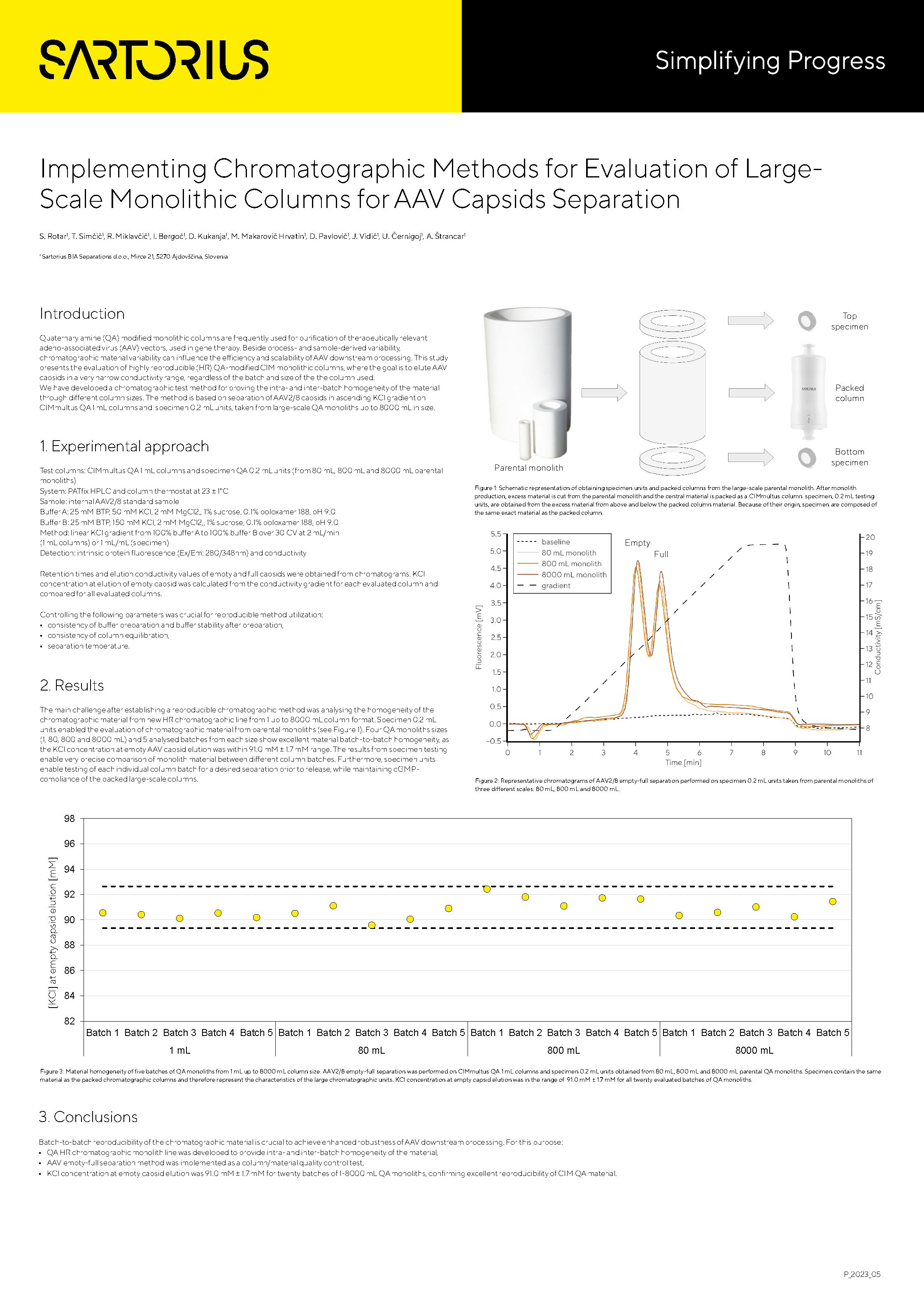
Implementing Chromatographic Methods for Evaluation of Large Scale Monolithic Columns for AAV Capsids Separation
Quaternary amine (QA) modified monolithic columns are frequently used for purification of therapeutically relevant adeno-associated virus (AAV) vectors, used in gene therapy. Beside process- and sample-derived variability, chromatographic material variability can influence the efficiency and scalability of AAV downstream processing.
This study presents the evaluation of highly reproducible (HR) QA-modified CIM monolithic columns, where the goal is to elute AAV capsids in a very narrow conductivity range, regardless of the batch and size of the column used. We have developed a chromatographic test method for proving the intra- and inter-batch homogeneity of the material through different column sizes. The method is based on separation of AAV2/8 capsids in ascending KCl gradient on CIMmultus QA 1 mL columns and specimen 0.2 mL units, taken from large-scale QA monoliths up to 8000 mL in size.