The PATfix pDNA analytical platform package was helpful in getting the plasmid analysis running with validated protocols built-in the system. We have also been utilising PATfix for the analysis of IVT mRNA with validated protocols provided by BIA Separations, Inc.. We have been getting consistent and reliable results with high-throughput work processes made possible by the at-line implementation of the PATfix platform. I am happy with the system as it is easy to use, reliable and the support provided by the highly knowledgeable team at BIA Separations, Inc. and Sartorius has been excellent. I am looking forward to continuing working with the PATfix platform and the BIA Separations, Inc. team with their ongoing developments in providing high-quality analytical products. – Dae Jong Han, Research Specialist (Analytics), BASE mRNA Facility, Australia.
Read moreLNP Process Analytics Services
In the versatile field of drug delivery and vaccines, lipid nanoparticles (LNPs) are particularly effective for nucleic acids, offering enhanced cellular uptake and controlled release. However, their complex structure presents significant challenges in characterization and analysis. Effective LNP evaluation requires thorough analysis of size, polydispersity, surface charge, stability, encapsulation efficiency, and nucleic acid integrity. Advanced techniques, such as liquid chromatography, are essential for ensuring LNP quality and efficacy. BIA Separations, Inc. experts ensure the stability, integrity, and consistency of LNP formulations by enabling comprehensive analysis and quality control.
Common Challenges in LNP Analytics
- Sample Pre-Treatment Required: LNPs must be dissolved or disassembled and pre-treated before chromatographic analysis, leading to potential sample loss, contamination, and increased processing time.
- Label Dependency: Using labels in analytical processes risks unwanted background signals from non-specific binding and limits the scope of analysis, preventing kinetic and binding specificity assessments.
- LNP Instability: Lipid structure affects particle stability, with oxidable lipids inducing mRNA oxidation, ionizable lipids improving transfection efficiency, and helper lipids like cholesterol, DSPC, or DOPC contributing to stability and rigidity.
- CQA Determination Challenges: Meeting CQA requirements for LNPs relies on the presence of specific constituents, such as polyethylene glycol (PEG)-lipids, which define LNP physicochemical characteristics and biological activity.
- Encapsulation Efficiency Limitations: Current methods for determining encapsulation efficiency lack additional information about therapeutically relevant parameters, such as mRNA integrity.
With Cornerstone services, these common challenges in LNP characterization and analysis are addressed, providing a solution tailored to specific needs.
Overcoming LNP Analytical Challenges
The innovative PATfix LNP Switcher significantly improves traditional assays. Using a 2D chromatographic method, it characterizes LNP formulations in a single run without sample preparation. It quantifies encapsulated and naked nucleic acid cargo, estimates mRNA fragmentation, and determines LNP size.
- Direct Injection Capability
Enables direct injection of LNP formulations, allowing simultaneous separation and quantification of lipid components and nucleic acids without sample pre-treatment. - UV-Based Detection
Relies solely on UV response, eliminating the need for dyes.
- Lipid Composition Monitoring
The method tracks changes in lipid composition throughout the LNP manufacturing process, ensuring stability and consistency of LNP formulations.
- Versatile Quality Control
Applies to various LNP formulations, robustly detecting differences in lipid ratios and mRNA, making it a critical quality control tool for LNP-based therapeutics.
- Encapsulation Efficiency Analysis
PATfix LNP Switcher determines encapsulation efficiency based on an absorbance signal, allowing independent assessment of integrity for both encapsulated and non-encapsulated nucleic acids.
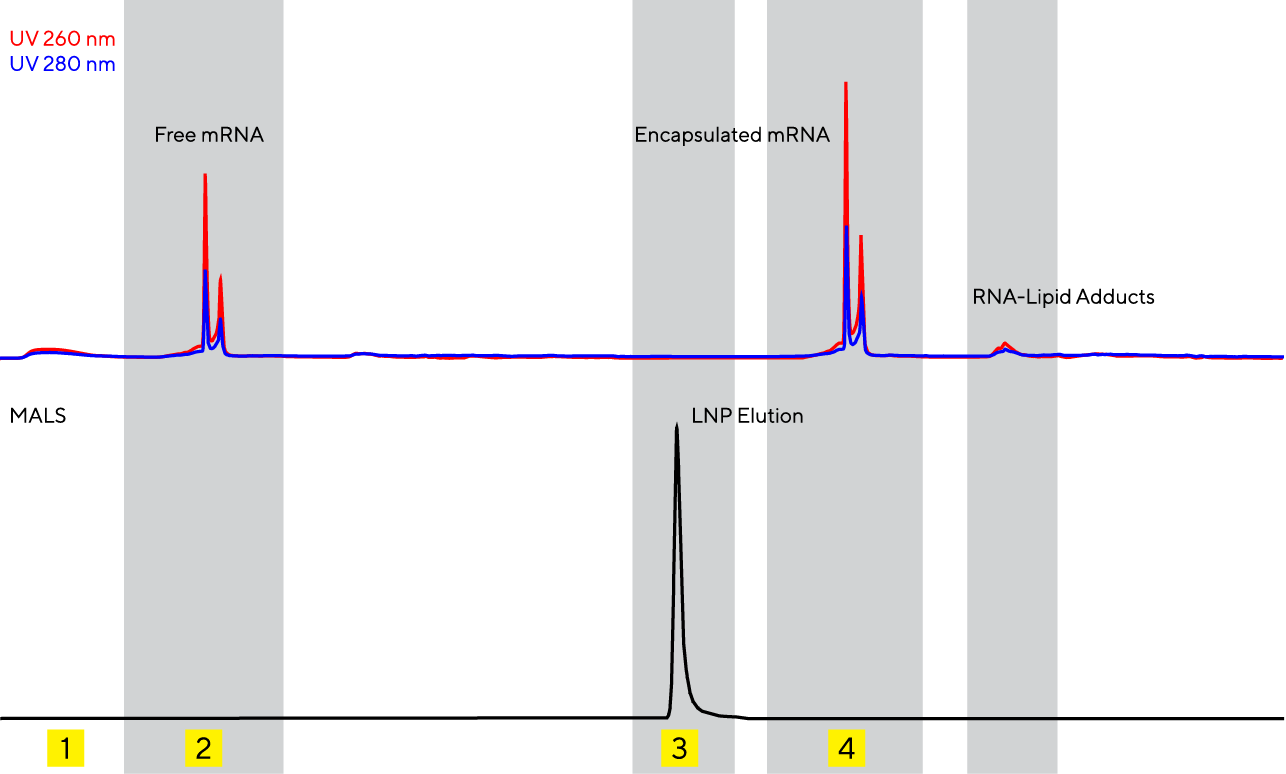
- Load:
Direct injection of LNP formulations; no sample pre-treatment, only dilution with loading buffer
- Analysis of Free Nucleic Acids:
HIC (hydrophobic interaction chromatography) column with large 6 μm channels allows for the selective binding of formulated LNPs and separation of free nucleic acids. Free nucleic acids are separated on the CIMac SDVB column, and analyzed for quantity, size and integrity.
- LNP Elution:
The elution of LNPs is detected by the MALS detector, where size and heterogeneity of particles is determined.
- Analysis of Encapsulated Nucleic Acids:
Reverse phase analytics enables both LNP deformulation and payload release in one step. Encapsulated nucleic acid can be analysed using UV-Vis detector for multiple parameters: concentration, type, size, integrity as well as RNA-lipid adducts.

- Load:
Direct injection of LNP formulations; no sample pre-treatment, only dilution with loading buffer
- Analysis of Free Nucleic Acids:
HIC (hydrophobic interaction chromatography) column with large 6 μm channels allows for the selective binding of formulated LNPs and separation of free nucleic acids. Free nucleic acids are separated on the CIMac SDVB column, and analyzed for quantity, size and integrity.
- LNP Elution:
The elution of LNPs is detected by the MALS detector, where size and heterogeneity of particles is determined.
- Analysis of Encapsulated Nucleic Acids:
Reverse phase analytics enables both LNP deformulation and payload release in one step. Encapsulated nucleic acid can be analysed using UV-Vis detector for multiple parameters: concentration, type, size, integrity as well as RNA-lipid adducts.
Why Choose Cornerstone LNP Process Analytics Services?
By partnering with Cornerstone LNP Process Analytics Services, the following benefits are offered:
- Direct injection of LNP formulations for simultaneous separation and quantification of nucleic acids.
- Comprehensive evaluation of payload integrity and LNP size distribution.
- Label-free analysis using UV and MALS responses.
- Continuous monitoring of lipid composition changes during storage, manufacturing, and purification.
- Enhanced LNP stability and consistency.
- Accurate detection of variations in lipid ratios and mRNA.
- Robust quality control for LNP-based therapeutics.
Additional Resources

Read the article Quantitative Analysis of Lipids and Nucleic Acids in Lipid Nanoparticles Using Monolithic Column for a more detailed insight.

Read the article Quantitative Analysis of Lipids and Nucleic Acids in Lipid Nanoparticles Using Monolithic Column for a more detailed insight.
Hear From Our Customers
Ready to Discuss Your Project?
FAQ About Services for LNP Process Analytics
Our facilities are located in Wilmington, Slovenia. We can support you with process development in our modern, fully-equipped BSL 1 and 2 facilities. Take a virtual tour of our laboratories.

Yes, the reverse-phase column’s capability lies in its ability to separate species by size and hydrophobicity. This enables the detection of smaller payload species, representing fragmented payload, as well as lipidated adducts.
With this analytical setup, pDNA up to 15 kbp and mRNA up to 10 kb can be analyzed.
Yes, the same method is used for the analysis of liposomes and other lipid particles. This system configuration allows for straightforward optimization of the chromatographic method to suit the analysis of unique lipid particles.
Consult Our Experts
Our experts would be happy to discuss your project. Fill the contact form below or send us an email to support@biaseparationsinc.com





