The PATfix pDNA analytical platform package was helpful in getting the plasmid analysis running with validated protocols built-in the system. We have also been utilising PATfix for the analysis of IVT mRNA with validated protocols provided by BIA Separations, Inc.. We have been getting consistent and reliable results with high-throughput work processes made possible by the at-line implementation of the PATfix platform. I am happy with the system as it is easy to use, reliable and the support provided by the highly knowledgeable team at BIA Separations, Inc. and Sartorius has been excellent. I am looking forward to continuing working with the PATfix platform and the BIA Separations, Inc. team with their ongoing developments in providing high-quality analytical products. – Dae Jong Han, Research Specialist (Analytics), BASE mRNA Facility, Australia.
Read moremRNA Process Analytics Services
In biopharmaceutical research, mRNA analytics are essential for the development of mRNA-based therapies and vaccines. Accurate mRNA analysis ensures quality, efficacy, and safety. Comprehensive analytics evaluate mRNA quantity, integrity, purity, and capping efficiency, optimizing manufacturing for scalability and reproducibility. Understanding mRNA analytics is crucial for meeting regulatory standards and accelerating the development of novel treatments. BIA Separations, Inc. offers advanced methods to address key quality attributes of mRNA.
Common Challenges in mRNA Analytics
- IVT Optimization and mRNA Production Monitoring: Ensuring maximum utilization of NTPs to produce target mRNA in the IVT reaction and identifying the point at which mRNA production ceases.
- mRNA Stability and Fragmentation Analysis: Ensuring mRNA integrity during production to maintain vaccine efficacy by optimizing conditions for stable, high-quality mRNA and preventing fragmentation.
- Capping Quality: Ensuring proper mRNA capping to optimize translation and enhance immune response activation in vaccines, verifying capping quality for vaccine effectiveness.
Advanced Cornerstone® services for mRNA analytics tackle common production challenges, offering solutions specifically designed to meet specific needs.
Overcoming mRNA Analytical Challenges
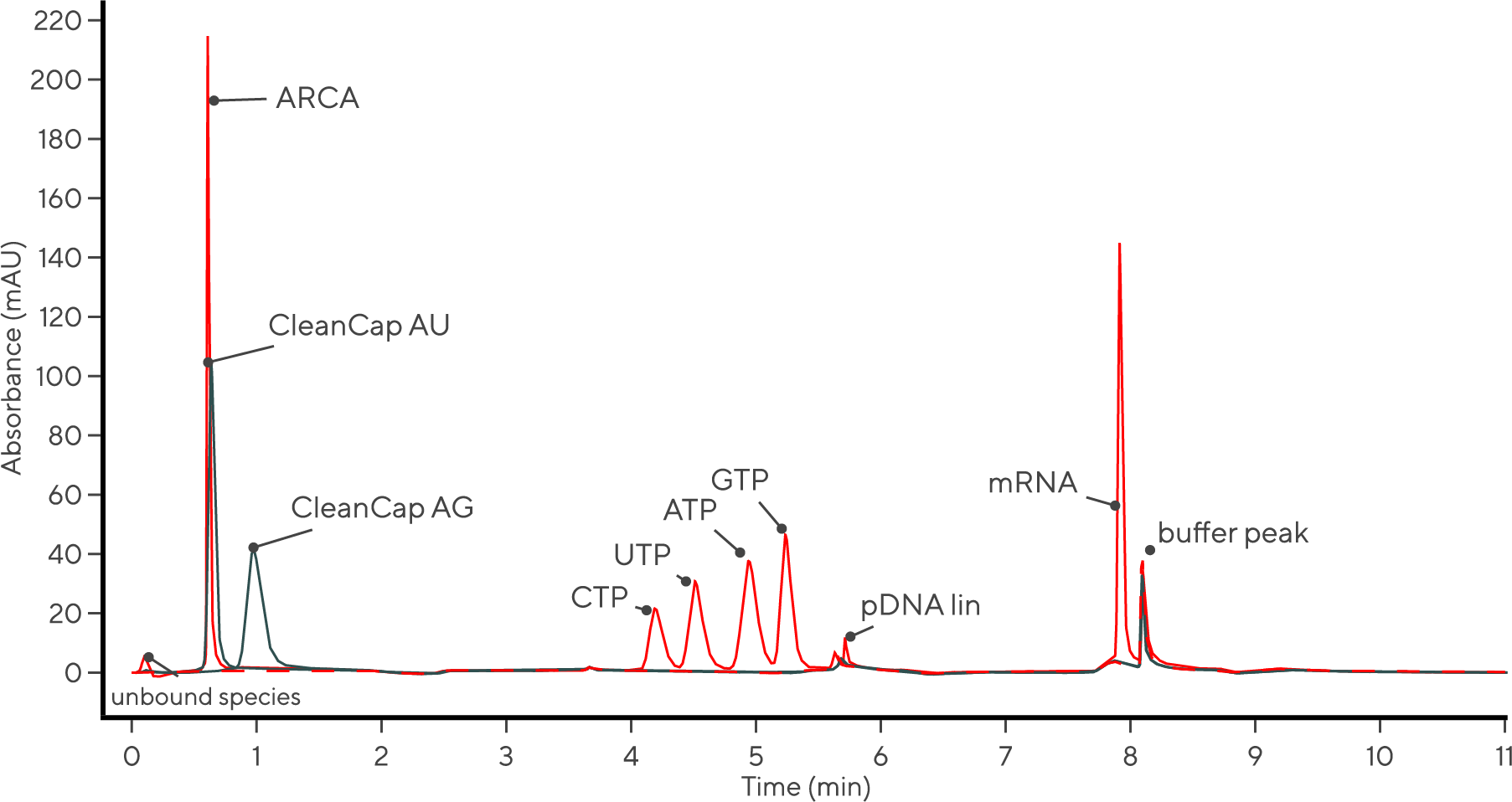
The integration of PATfix® analytics and complementary methods, including capillary gel electrophoresis (CGE), delivers a comprehensive evaluation of mRNA samples. Our analytical methods and the PATfix® mRNA platform address key quality attributes of mRNA, providing reliable at-line insights during process development and production, focusing on IVT reaction components and final product quality control.
- Quantitative IVT Monitoring
The IVT monitoring method enables quantitative at-line monitoring of individual NTP usage and mRNA synthesis. This optimization of the NTP ratio, along with modified NTPs and determination of the optimal IVT run-time, makes the process more cost- and time-effective. Additionally, it supports fed-batch IVT reactions, increasing yield and reducing costs.
- mRNA Fragmentation Analysis
Monolithic chromatography is used to determine the extent of mRNA fragmentation during production and purification steps. - Capping Quality Assessment
The method enables fast, reliable, and inexpensive determination of the percentage of capped mRNA.
Why Choose Cornerstone mRNA Process Analytics Services?
By partnering with Cornerstone mRNA Process Analytics Services, the following benefits are offered:
- Quantitative IVT monitoring for precise NTP usage and mRNA synthesis.
- Optimized NTP ratio as well as modified NTP ratio, and IVT run-time for cost and time efficiency.
- Support for fed-batch IVT reactions, increasing yield and reducing costs.
- mRNA fragmentation analysis during production and purification.
- Fast, reliable, and cost-effective capping quality assessment.
Additional Resources
Read these articles for a more detailed insight:
Read these articles for a more detailed insight:
-
Gram-Scale mRNA Production Using a 250-mL Single-Use Bioreactor
-
mRNA Fragmentation and Quality Assessment Using Ion Pair Reverse-Phase Analytics
Hear From Our Customers
Ready to Discuss Your Project?
FAQ About Services for mRNA Process Analytics
Our facilities are located in Wilmington, Slovenia. We can support you with process development in our modern, fully-equipped BSL 1 and 2 facilities. Take a virtual tour of our laboratories.

Using the mRNA platform, you can monitor mRNA production during the IVT reaction, determine mRNA concentration, and assess the mRNA integrity and capping efficiency.
All these different RNA species can be analyzed using our chromatographic method. Some individual optimization may be necessary to achieve the best possible results.
The mRNA standard for the system suitability test is available and can be ordered here (Cat.no.: BIA-mFix4.1.1).
We offer mRNA testing using capillary gel electrophoresis as an orthogonal method to chromatographic evaluation.
Consult Our Experts
Our experts would be happy to discuss your project. Fill the contact form below or send us an email to support@biaseparationsinc.com






