Key Differences Between CIM QA and CIM QA HR
In biomolecular purification, achieving high reproducibility and purity is vital, especially for AAV Empty-Full separation. As the demand for gene therapies continues to rise, the need for advanced chromatographic solutions like CIMmultus QA HR (High Reproducibility) products are becoming increasingly vital.
In this blog post, we’ll take a closer look at the specific functionalities and release testing criteria of CIMmultus QA and CIMmultus QA HR. Whether you’re optimizing your process or ensuring high reproducibility, understanding these columns’ roles can significantly impact your success in AAV purification.
Overview of the Chemistry and Applications
Both CIM QA HR and CIM QA are quaternary amine-based anion exchangers, sharing the same chemistry backbone. The CIM QA product line was developed for a wide range of biomolecules (such as adenovirus, lentivirus, VLP, bacteriophage, influenza virus etc.). Whereas CIM QA HR line offers distinct advantages tailored for sensitive AAV Empty–Full separation, addressing industry demands.
CIM QA HR allows consistent purity in the enrichment of full AAV capsids, regardless of batch or product scale. On the other hand, CIM QA serves as a suitable option for AAV polishing if there is no need for a step gradient, and the linear gradient provides sufficient conductivity tolerance in the process, allowing for some variability in the separation of empty and full AAVs.
CIM QA and CIM QA HR perform similarly in terms of the resolution of Empty-Full separation. The differences between them arise from different release criteria, leading to different reproducibility performance.
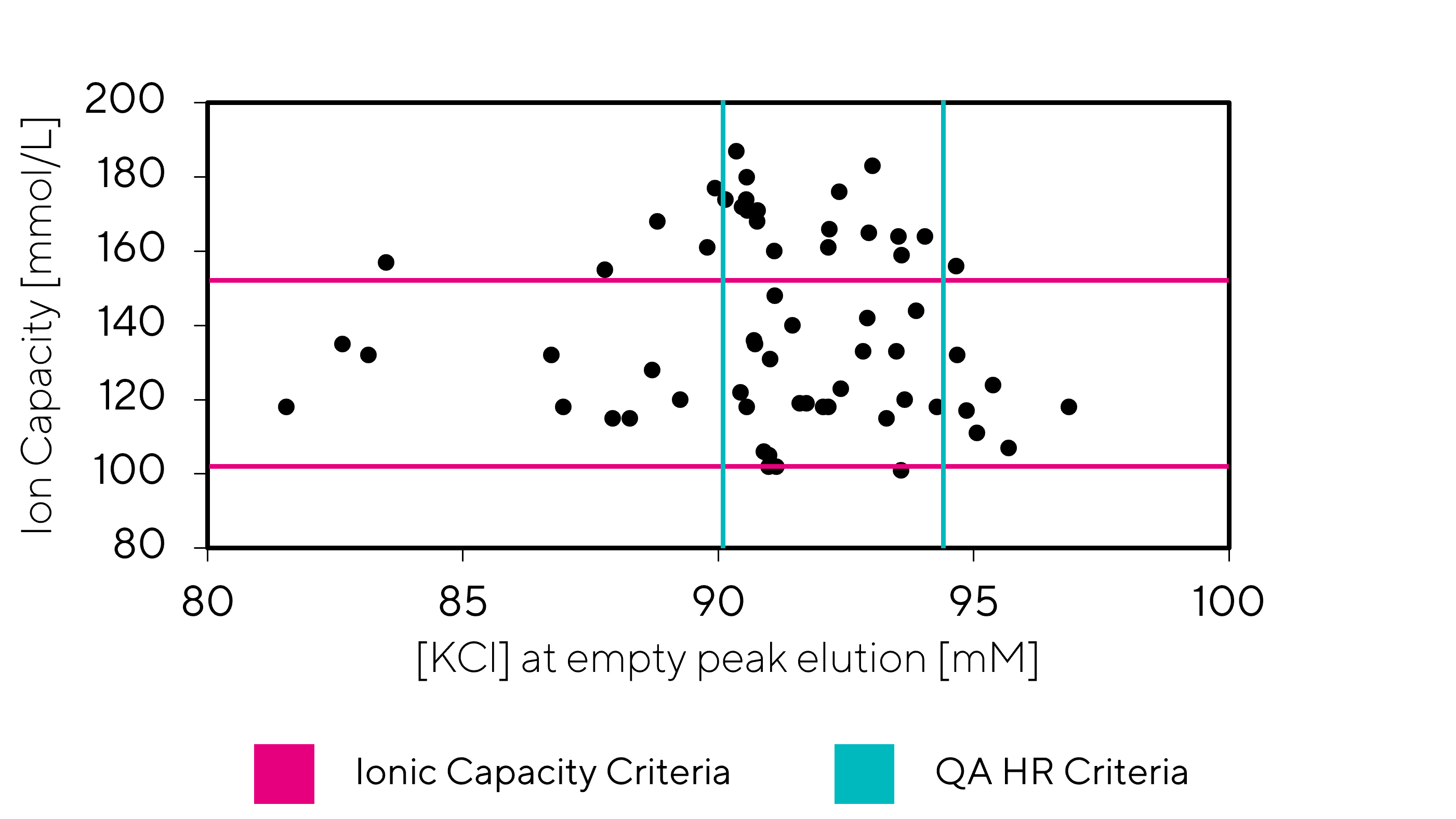
The CIM QA monoliths are selected based on the Ion capacity release test, which is dependent on the capacity for small molecules and is less suitable for the delicate process of AAV Empty-Full separation. However, they are designed for widespread use across various types of biomolecules.
In contrast, the release test for the CIM QA HR line is based on the results of AAV E/F separation performed in an ascending linear KCl elution gradient. Monoliths are selected based on performance with a tolerance of +/- 3% conductivity of the empty AAV elution peak. AAV elutes at a specific KCl concentration within the linear gradient, regardless of the CIM QA HR monolith volume. This approach ensures high reproducibility across the entire scale and across different CIM QA HR product lines.
Testing and comparing several CIMmultus QA and CIMmultus QA HR columns has proven that there is no correlation between the tests for release criteria described in the previous paragraph. A representative graph of the release criteria is shown on Figure 1, where each dot represents the results of an individual column.
Amine ligands, especially the QA, are susceptible to hydrolysis in exposure to alkaline conditions. Due to the narrow release criteria of CIM QA HR, the cleaning-in-place (CIP) solutions have also been adapted. Column lifetime and performance can significantly decrease if the product is not properly cleaned and neutralized (to displace the OH ions) before and during the storage. Recommended cleaning, neutralization, and storage solutions for CIM QA HR have been adjusted to achieve higher reproducibility when the product is reused for AAV Empty-Full separation.
Both QA and QA HR monoliths are available in a wide range of CIM formats, CIMac for analytical purposes, CIM Monolithic Well Plates and CIM Octa Columns, which allow cost-effective fast screening and high throughput. CIMmultus format of QA and QA HR is designed for preparative purposes, ranging from R&D scales (1, 4, and 8 mL) up to large industrial scales (up to 40 L columns). CIMmultus columns are available as cGMP and non-cGMP. cGMP QA HR columns feature a Specimen unit, which has the same characteristics as parental column characteristics as parental column and it is primarily utilised as a control to follow column lot-to-lot variability.
CIM QA HR: Seamless Gradient Transition and Superior Reproducibility
CIM QA HR monoliths offer the advantage of seamless transition from a linear to step gradient, ensuring that full capsids consistently elute at the same salt concentration across various scales, highlighting superior performance and reproducibility.
Overview of Differences
| QA | QA HR | |
| Ligand | Quaternary amine | Quaternary amine |
| Purpose | Virus purification (e.g. LV, Influenza virus …), protein purification, ribosomes purification, etc. | Highly sensitive separations, such as AAV E/F separation or EV purification |
| QC testing | Ion capacity testing with small molecules | AAV test using ascending KCl elution gradient with internal AAV standard. The selection is based on the narrow acceptable interval (+/- 3%) of KCl concentration required for elution of empty AAV capsid peak at defined chromatography conditions |
| Housing and column dimensions | Traditional CIMmultus housing | 1 mL, 4 mL and 8 mL columns have adjusted monolith dimensions and housing |
| Recommended cleaning and sanitisation solutions | 1 M NaOH + 2 M NaCl | 0.1 NaOH + 2 M NaCl for cleaning 0.5 NaOH + 2 M NaCl for sanitisation |
| Storage solution | 20% Ethanol | 20% Ethanol in 5 mM sodium phosphate at pH 6.0 |
| Shelf life | 5 years | N.D. |
| Availability of CIMmultus cGMP and non-cGMP column formats | non-cGMP: 1 mL – 8 L
cGMP: 4 mL – 40 L |
non-cGMP: 1 mL – 8 mL
cGMP: 4 mL – 40 L |
| Specimen unit | No | Yes, for cGMP units |
Related Posts
A Guide to Monolithic Column Channel Sizes
A guide to choosing the right channel size of monoliths for biomolecules: pDNA, mRNA, AAV, LVV, bacteriophages, LNP, proteins, and more
What’s Holding Back mRNA-LNP Therapeutics?
Solving challenges in the areas of purity, consistency, and monitoring technologies is crucial for the development of mRNA-LNP therapeutics.
How Acidic Denaturation Facilitates dsRNA Removal
How to remove double stranded RNA for safer mRNA therapeutics, without special equipment and preserving the target molecule
The Next Generation of Chromatography: Moving From Classic HPLC to Advanced Platforms
Advanced chromatography platforms enhance process development with integrated solutions beyond traditional HPLC systems.




